COVID-19 ANTIBODY FACT SHEET
This COVID-19 Antibody fact sheet contains information to help you understand the risks and benefits of using the COVID-19 IgG testing provided by EDP Biotech Corporation. After reading this Fact Sheet, if you have questions or would like to discuss the information provided, please talk to your healthcare provider.
It is important that if you are experiencing difficulty breathing and/or fever that you contact your healthcare provider as soon as possible.
For the most up to date information on COVID-19, please visit the CDC Coronavirus Disease 2019 (COVID-19) webpage: https://www.cdc.gov/COVID19
What is COVID-19?
COVID-19 is caused by the SARS-CoV-2 virus. The virus, which can cause mild to severe respiratory illness was first identified in Wuhan, China and now has spread globally. There is limited information available to characterize the spectrum of clinical illness associated with COVID-19, but it likely spreads to others when a person shows signs or symptoms of being sick (e.g., fever, coughing, difficulty breathing, etc.)
What is the COVID-19 IgG Antibody Test?
The COVID-19 IgG tests provided by EDP Biotech Corporation are designed to detect antibodies against the virus that causes COVID-19 in blood serum specimens. These tests are currently going through the FDA approval process under an Emergency Use Authorization (EUA) application. This process allows the use of these tests while the application is under review by the FDA.
Why should I get an antibody test?
The focus of the COVID-19 IgG tests are to help determine if you may have been exposed and potentially have some protection to the virus that causes COVID-19. These blood test results are to be used for screening purposes only. These blood tests results are not to be used for diagnostic purposes.
Where can I get a test?
If you need a test for yourself or your family we offer those at our lab or at a preferred partner location. Visit our Tests for Individuals page. If you are an Employer, we offer on-site services to efficiently test your whole team. See services for businesses. If you provide health or wellness services you can administer EDP COVID-19 test to your patients. Visit the Services for Physicians page.
What is the Clinical Performance of this test?
EDP Biotech’s CLIA laboratory currently utilizes the serological assay developed by trusted partner Quansys Biosciences in Logan, Utah. The developer submitted for an EUA with the FDA on May 14th, 2020. The results from their Clinical Agreement Validation N=576 sample submission are shown below:
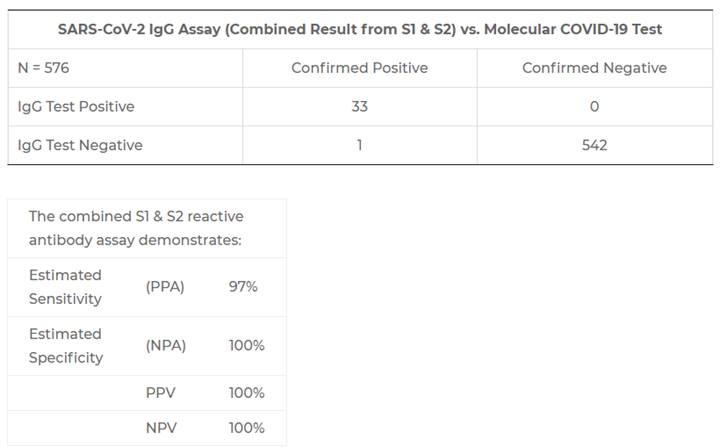
In-House Testing Results
Results of EDP Biotech’s in-house validation studies are shown below, confirming the Quansys Biosciences test performance in our Knoxville CLIA laboratory:
Clinical Agreement Study
The comparator method used to establish clinical truth is the Abbott RealTime SARS-CoV-2 RT-PCR test or samples collected prior to November 2019.
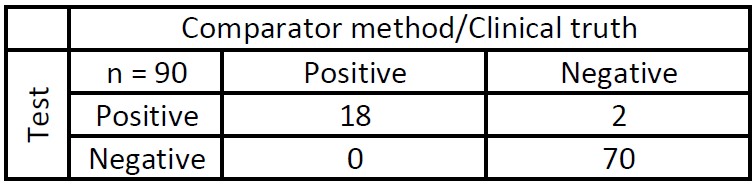
Sensitivity (Positive Predictive Agreement) 100%
Specificity (Negative Predictive Agreement) 97%
Acceptance Criteria PASSED
What do the results mean?
Positive Result:
A positive result means it may be likely that you have COVID-19 and that you may need isolation to avoid spreading the virus to others. Other results may indicate you were infected previously. Please contact your medical provider to determine how best to care for you based on the test results along with other factors. There is also a chance of false-positive results, meaning that this test can give a positive result that is incorrect.
Negative Result:
A negative test result means that the antibodies to the virus that causes COVID-19 were not found in your sample. However, it is possible for this test to give a false negative result, meaning that this test can give a negative result that is incorrect. If you are tested early in your illness and antibodies haven’t been produced, then a negative result may occur. This means that it is possible you could still have COVID-19 even though the test result is negative. Your healthcare provider will consider the test result together will all other aspects of your medical history in deciding how to care for you.
Order Your Antibody Test
Reproducibility
Five samples tested on five separate days for agreement.
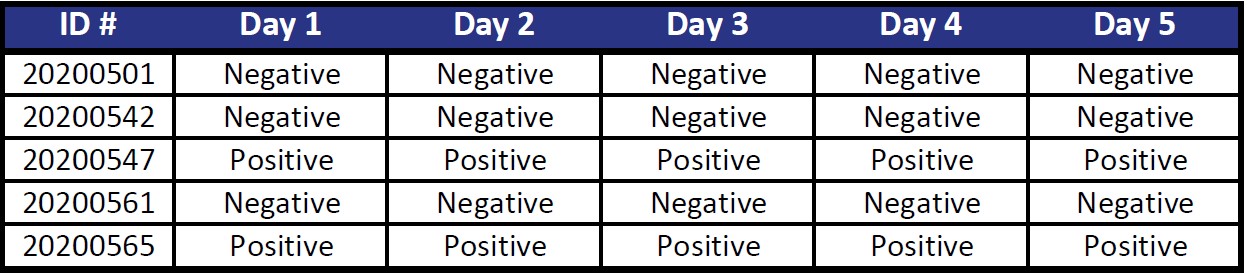
Percent Agreement 100%
Acceptance Criteria PASSED (Agreement >= 95%)
Cross-Reactivity
Five samples collected prior to November 2019 from each of the following disease states were tested for cross-reactivity to SARS-CoV-2 spike 1 (S1) and spike 2 (S2) antigens. Results are only considered positive when reactive to both S1 and S2.
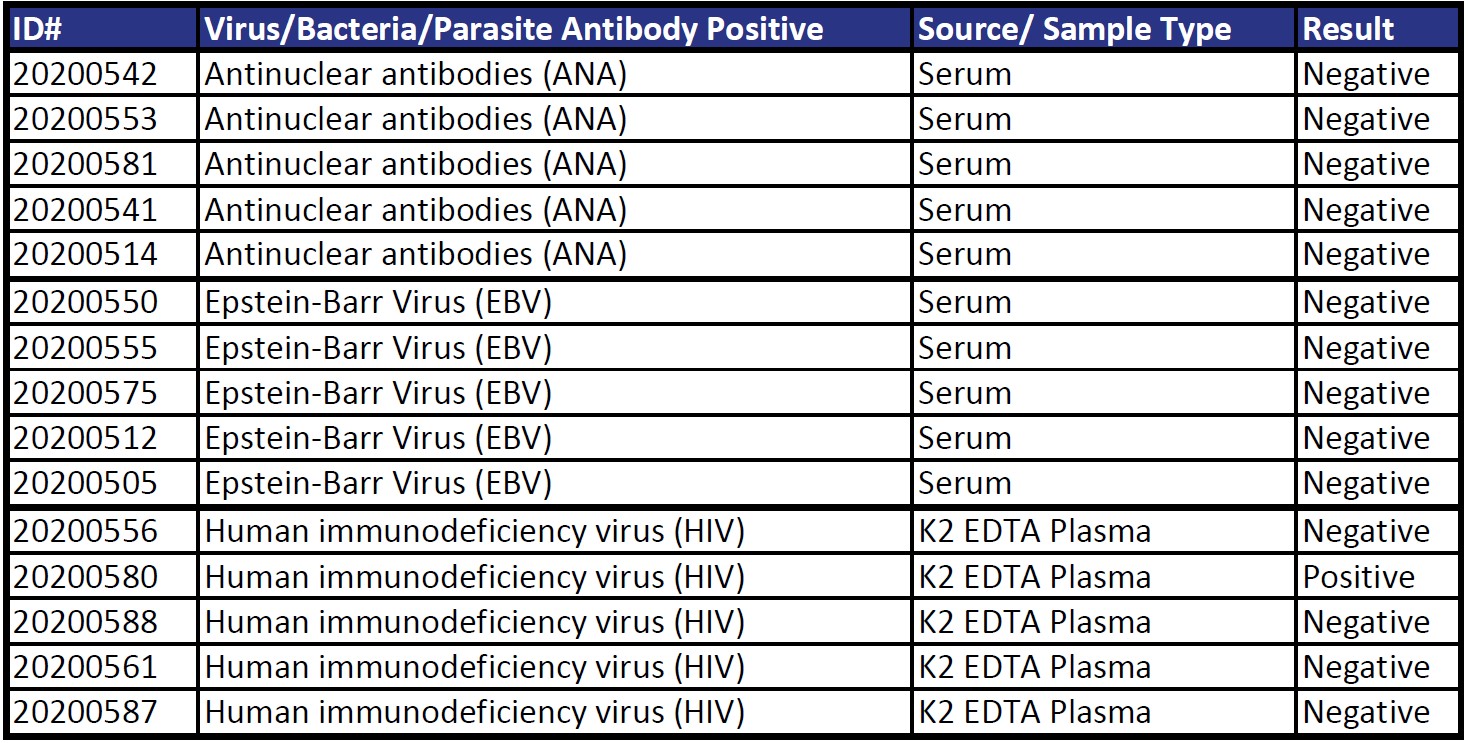
Total Percent Agreement (n = 90) 98%
Acceptance Criteria PASSED
Matrix Equivalency
Each possible matrix type collected at the same time from ten individuals and tested for agreement.
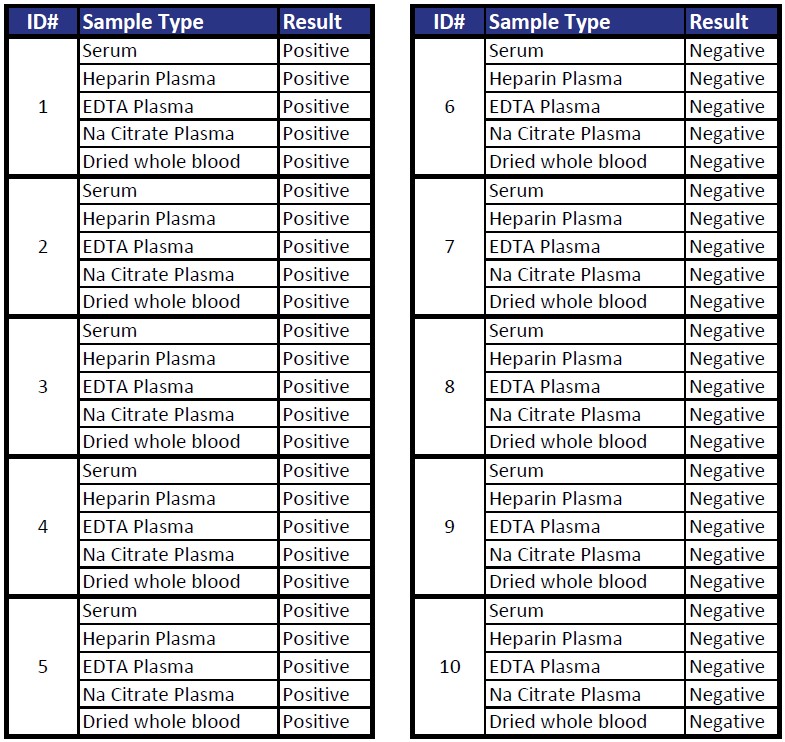
Percent Agreement 100%
Acceptance Criteria PASSED
